Alzheimer’s Brains Mottled with Epigenetic Changes
Quick Links
Epigenetic modifications control gene expression, but scientists still don’t know if or how they contribute to disease. To address this knowledge gap, the National Institutes of Health launched the Roadmap Epigenomics Project in 2008 to compare epigenomes in healthy and diseased cells. In the August 17 Nature Neuroscience, two papers from separate but collaborative research groups report on some of the fruits of that effort. Both groups surveyed DNA methylation in hundreds of human AD and control brains and identified several regions where changes in this epigenetic mark correlated with the amount of Alzheimer’s pathology. The results may help flag genes that are turned up or down in AD, and provide insight into pathogenesis, said Philip De Jager at Brigham and Women’s Hospital, Boston, the first author of one of the papers. Some of this data was previously presented at the 2012 Society for Neuroscience conference (see Nov 2012 conference story).
“For perhaps the first time, our papers find robust evidence for epigenetic changes in the brains of people with AD,” said Jonathan Mill at King’s College London, who led the other study. Notably, the two groups obtained largely overlapping sets of genes, although they used different cohorts and methods. De Jager told Alzforum, “To my understanding, this is the first time that two independent epigenetic studies have cross-replicated each other. It validates this approach to studying Alzheimer’s and brain diseases in general.”
Commentators called the data a promising advance, noting that much more work remains to understand how these changes affect the course of the disease. “These are well-designed and well-executed studies, and the data are convincing and exciting,” said Li-Huei Tsai at the Massachusetts Institute of Technology in Cambridge. “That these methylation changes correlated with plaque load, but not necessarily cognitive state, is also intriguing, indicating that these [modifications] occur early [in disease course].”
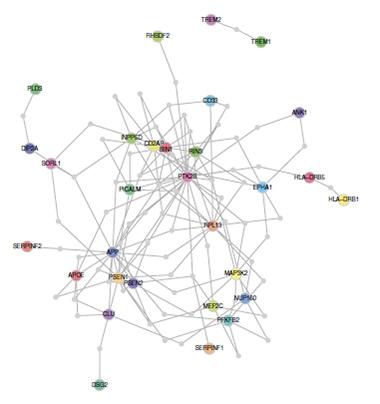
In Alzheimer’s brains, methylation differences change the expression level of the genes ANK1, RHBDF2, CDH23, RPL13, DIP2A, SERPINF1, and SERPINF2. Network analysis links six of these genes to known AD risk factors. [Image courtesy of De Jager et al., Nature, 2014]
DNA methylation adds a methyl group to a cytosine base, usually one located next to a guanine in what is called a CpG site. This modification changes the expression level of nearby genes. Previous research hinted at a role for methylation, as well as other epigenetic modifications, in AD (see Mar 2007 news story, Jul 2008 news story, Aug 2009 news story, and Mar 2012 news story). However, most older studies were small and findings often conflicted, with some showing an overall decrease in methylation in AD and others an increase. The Roadmap project sought to clarify the picture by funding four large surveys of methylation in AD brains (see Jun 2010 conference series).
One study, led by De Jager and David Bennett at Rush University, Chicago, examined postmortem prefrontal cortexes from more than 700 participants in the Rush Memory and Aging Project and the Religious Orders Study. Around 60 percent of the brains met pathological criteria for AD, but only about two-thirds of those had been clinically diagnosed with the disease; the others were cognitively normal or mildly impaired. The authors determined methylation status with a commercial CpG beadchip array, in which different probes bind to methylated versus unmethylated sequences. The chip included half a million CpG sites, about 5 percent of the total in the human genome. The authors correlated methylation at each of these sites with the amount of neuritic amyloid plaques in the brain.
In DNA extracted from these cortical tissues samples, the authors found 71 motifs where methylation changed in tandem with amyloid-β neuritic plaque burden. DNA was more methylated at about 80 percent of these sequences and less so at the other 20 percent. The authors calculated that methylation changes at all of these sequences would explain about 29 percent of the plaque burden in this cohort. That was greater than the effect of all known AD genes, which accounted for just 14 percent of the plaque burden in this group, they authors claim. Two methylation sites fell near the known AD risk genes BIN1 and ABCA7, currently second and fourth on AlzGene Top Results. To confirm the findings, the authors tested all 71 motifs in an independent cohort of 117 brains. Eleven correlated with plaque load; many of the other sites showed trends, suggesting they might reach statistical significance in a larger cohort, the authors noted.
How do these methylation changes affect gene expression? The answer is not trivial, since methylation can affect genes some distance away and has variable effects on expression, depending on where it occurs. Hypermethylation near the start of a gene usually silences expression, but near the end of a gene can turn it up, De Jager said. To investigate, the authors measured RNA levels from 21 genes near the 11 CpG sites in an independent cohort from the Mayo Clinic. In AD brains they found seven genes whose expression was altered. None were known AD genes, but using the Disease Association Protein-Protein Link Evaluator (DAPPLE), an algorithm that analyzes reference datasets of protein-protein interactions, the authors found that six of the genes had close ties to Alzheimer’s genes. Three of them—DIP2A, SERPINF1, and SERPINF2—interact with proteins involved in processing amyloid precursor protein, such as SORL1 and PSEN2, while three others—ANK1, RHBDF2, and CDH23—have links to microglia and astrocytes.
Many of the 71 CpG sites turned up in the other study, led by Mill and Leonard Schalkwyk at King’s College London. The authors used the same array De Jager used to measure methylation in four different brain regions from 122 donors in the MRC London Brain Bank for Neurodegenerative Disease. Mill and colleagues correlated methylation with Braak staging rather than neuritic plaques, but nonetheless obtained a set of methylation sites that largely overlapped with those in De Jager et al. Mill and colleagues replicated their findings in two independent brain cohorts. Methylation profiles were similar among three cortical regions examined (entorhinal cortex, superior temporal gyrus, and prefrontal cortex), but a distinct methylation pattern emerged in the cerebellum, a region typically spared in AD, and in whole blood. This suggests the observed methylation changes are specific to the cortex, Mill told Alzforum.
The top hit in this study was ANK1, a member of the ankyrin family of proteins that anchor membrane proteins to the cytoskeleton. ANK1 variants are associated with Type 2 diabetes, a risk factor for Alzheimer’s (see AlzRisk analysis). Alternative RNA splicing creates numerous isoforms of ANK1. Because the methylation site occurs upstream of a splice site, the authors looked for changes in isoform expression. They found that the relative abundance of three isoforms varied in brains with high AD pathology compared to low, though they did not correlate this with methylation status.
Together, these findings help broaden the understanding of Alzheimer’s pathways and highlight new genes for investigation, commentators agreed. “Those of us working on the epigenetics of AD are starting down a long road,” said Paul Coleman at Banner Sun Health Research Institute, Sun City, Arizona, who leads another Roadmap project. “These findings are an excellent start.” Future studies should pursue what methylation does to gene expression, Coleman suggested, adding, “In biology, gene expression is where the rubber meets the road.”
De Jager plans to examine other epigenetic modifications in the differentially methylated regions to develop a fuller picture of how nearby genes are turned up or down in AD. Based on such differential expression patterns, he will generate a protein interaction network model of Alzheimer’s. The Accelerating Medicines Partnership and the NIH recently funded this project (see Feb 2014 news story and Sep 2013 news story).
Both research groups intend to focus on which cell types control the association between methylation changes and worsening AD pathology. Does methylation at a particular site happen primarily in neurons, microglia, or astrocytes? Do these methylation changes increase pathology, or vice versa? If methylation is a cause, then pharmaceutical companies might put resources toward targeting therapies to specific CpG motifs. Some current drugs do affect methylation, for example the antihypertensive hydralazine. They act on all sites indiscriminately. “Epigenetic modifications are potentially dynamic and reversible, so pharmaceutical companies are interested in developing agents that can reverse epigenetic dysfunction. In cancer, epigenetic drugs are at a quite high stage of development,” Mill noted. Several have been approved by the FDA, such as the histone deacetylase inhibitors vorinostat and romidepsin.—Madolyn Bowman Rogers
References
News Citations
- SfN: Epigenetic Changes in Alzheimer’s and Cognitive Decline
- Heavy Methyl—DNA, Protein Modification Affect Memory, APP, and Tau
- Drifting Toward AD—Epigenetic Changes Linked to Disease
- Twin Study Suggests Epigenetic Differences in AD
- Does Epigenetic Modification by Aβ Offer New Take on Therapy?
- New Initiative AMPs Up Alzheimer’s Research
- NIH Funds Prevention Trials and Translational Studies
Conference Coverage Series Citations
External Citations
Further Reading
Primary Papers
- De Jager PL, Srivastava G, Lunnon K, Burgess J, Schalkwyk LC, Yu L, Eaton ML, Keenan BT, Ernst J, McCabe C, Tang A, Raj T, Replogle J, Brodeur W, Gabriel S, Chai HS, Younkin C, Younkin SG, Zou F, Szyf M, Epstein CB, Schneider JA, Bernstein BE, Meissner A, Ertekin-Taner N, Chibnik LB, Kellis M, Mill J, Bennett DA. Alzheimer's disease: early alterations in brain DNA methylation at ANK1, BIN1, RHBDF2 and other loci. Nat Neurosci. 2014 Sep;17(9):1156-63. Epub 2014 Aug 17 PubMed.
- Lunnon K, Smith R, Hannon E, De Jager PL, Srivastava G, Volta M, Troakes C, Al-Sarraj S, Burrage J, Macdonald R, Condliffe D, Harries LW, Katsel P, Haroutunian V, Kaminsky Z, Joachim C, Powell J, Lovestone S, Bennett DA, Schalkwyk LC, Mill J. Methylomic profiling implicates cortical deregulation of ANK1 in Alzheimer's disease. Nat Neurosci. 2014 Sep;17(9):1164-70. Epub 2014 Aug 17 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
National Institute on Aging
These companion manuscripts report results from two NIA projects supported through the NIH Roadmap Epigenomics Program. They represent an important milestone in Alzheimer’s disease research because they suggest that changes in the DNA methylome may play a role in AD onset and progression and they illustrate that integrating epigenomic data with genomic, RNA expression, neuropathology, and clinical data will be a powerful way to gain new mechanistic insights about the complex etiology of AD.
Make a Comment
To make a comment you must login or register.